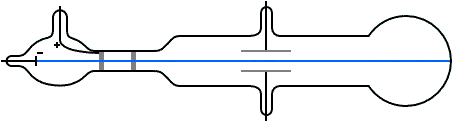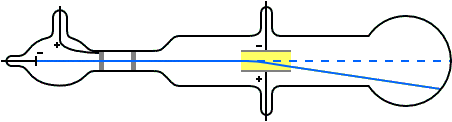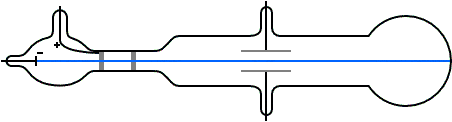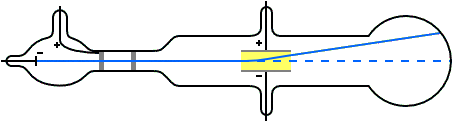
Instead, what flowed off the cathode toward the anode were called ‘cathode rays.’ Hence the name cathode ray tube. So, no one was calling it an electron beam. Thomson to see where the electron beam was hitting.Of course, before his experiment, we didn’t know electrons existed. On the opposite side of the tube is a coating that glows when struck by the electrons. Electrons, which have a negative charge, flow off the cathode and are attracted towards the anode.Ī small hole in the anode allows some electrons to pass through it, creating a beam of electrons. The cathode is a negatively-charged conductor, and the anode is a positively-charged conductor. On one side of the inside of the tube there’s a cathode and an anode. The CRT consists of several elements starting with a tube that’s vacuum sealed to keep air out of it. Thomson would’ve used, as seen in the diagram below. Thomson used the cathode ray tube to discover the electron, we need to know how a cathode ray tube works. Thomson to discover the existence of electrons in 1897.īefore we see how J.J.
It was cathode ray tubes that allowed the English physicist J.J. However, cathode ray tubes have been used for more than entertainment. The CRT in a television is used to display images on your screen. You might have used a cathode ray tube even if you’ve never even heard of it until reading this lesson.īefore LCD and Plasma TVs became commonplace, most people used bulkier cathode ray tube (CRT) televisions. This photograph is in the public domain.* All Partners were chosen among 50+ writing services by our Customer Satisfaction TeamĬathode Ray Tubes in Your Home Portrait of J.PBS atom builder References References for this article Measured masses and charges of the three elementary particles are given in the following table.For a neutral atom, number of proton=number of electron.Atomic mass = mass of protons + mass of neutron.

The gold foil experiment was originally conducted by Hans Geiger (left) and Ernest Marsden (right) under the supervision of Ernest Rutherford at the University of Manchester. James Chadwick (on the right), a student of Rutherford won Nobel Prize in Physics in 1935 for discovery of neutron. Ernest Rutherford (left) was awarded Nobel Prize in Chemistry in 1908 for his work in radioactivity. Here the symbol  \(_Z^XA\) is used where Z = atomic number and X = atomic mass of the element A. Figure 7.

Thomson measured the charge-by-mass-ratio (e/m) of cathode ray particle using deflection in both electric and magnetic field. Deflection of cathode rays towards positively charged plates Hence cathode ray consists of negatively charged particles. Applying electric field in the path of cathode ray deflects the ray towards positively charged plate. They are independent of the material composition of the cathode.ģ. The schematic of a cathode ray tube is given in figure 2. This ray is called cathode ray and the whole construction is called cathode ray tube. He detected that a stream of particle (ray) was coming out from the negatively charged electrode (cathode) to positively charged electrode (anode). Then he applied a high electrical voltage between two electrodes at either end of the tube. much of the air was pumped out of the tube. Thomson constructed a glass tube which was partially evacuated i.e. Thomson also won the Nobel Prize in 1937 for proving the wavelike properties of electron. Thomson won Nobel Prize in 1906 for discovering the elementary particle electron. Thomson in 1897 when he was studying the properties of cathode ray. Discovery of Electron Who discovered the electron?Įlectron was discovered by J.
